The 20 Year Term Rule
As a general rule of thumb, the effective life of a patent extends to 20 years from the application date. This rule has changed over time. For example, in the US, the rule was 17 years from the grant date prior to 1978. Some countries have different rules, but the major patent granting authorities, including the European Patent Office, Germany, China, Japan and Korea all follow the 20 year rule.
Should you assume that a patent is in force if its application was filed less than 20 years ago? Should you assume it is out of force if 20 years has passed? The answer to both questions is “No”. IFI CLAIMS® offers a convenient way to judge the status of granted patents. The IFI Patent Status Indicator is an easy-to-use data field that is calculated from expiration rules and subsequent legal status events. The Patent Status Indicator and IFI’s Adjusted Expiration Date are enhanced data fields available to all CLAIMS® Direct subscribers.
Note: IFI’s Adjusted Expiration Date and Patent Status Indicator are useful estimates, but should not be considered authoritative or final results. An attorney or legal service should be consulted to get an authoritative expiration and status determination for a granted patent.
Patents that expire before their 20th birthday
A patent can go out of force before its 20 year anniversary for several reasons—most commonly, because patent annuity fees have not been paid. IFI’s patstat patent Legal Status Indicator tracks these states for granted patents:
- Active
- Active - Reinstated
- Expired - Lifetime
- Expired - Fee Related
- Expired
- Withdrawn - After Issue
- Ceased
- Revoked
- In-force (EP Designated States only)
- Not-in-force (EP Designated States only)
Some of these states apply only to specific patenting authorities. IFI CLAIMS currently publishes the legal status indicator for the US, EPO, German, Canadian, Japanese, UK, French and Spanish patent offices. We are working to expand our coverage to other countries.
The figure below shows the patstat indicator for granted US patents with application dates in the year 1998. They are all less than 20 years old.
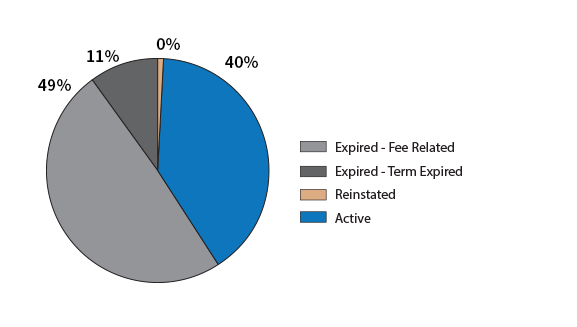
Patent Status of US Grants with Application Dates in the year 1998
You can see that 49 percent have expired due to lack of fee payments: they have been “abandoned”. This compares to 40% which are still active. 11% have expired because their lifetime has been exceeded. In the US, the 20 year rule applies to the first application. These patents may be continuations of pre-1998 applications. Less than 1% are in the small category of patents kept alive due to a provision that allows missed payments to be made retroactively.
The graph below shows how status changes over time. The current legal status of US grants with application dates from 1996 to 2016 are shown as percentages. Note that “Active” patents reach 50% in 2001.
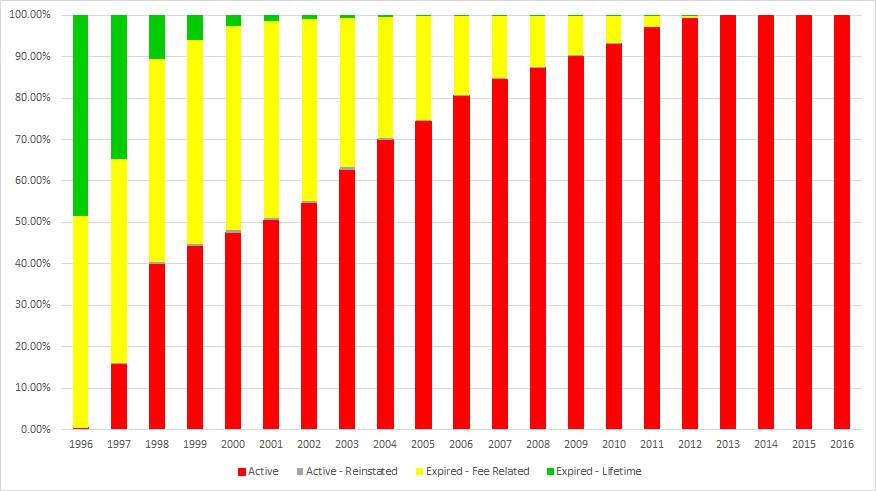
Percentage of patents in each expiration state based on application year
Once a patent reaches the “Expired – Fee Related” state, IFI only updates that status if the patent is reinstated. So, these patents never reach the “Expired – Lifetime” state. So, many of the patents applied for prior to 1998 would be “Expired – Lifetime” if they had not earlier expired for fee related reasons.
Patents that live past their 20th birthday
The data above shows that a patent can go out of force before its lifetime expiration date. But, a patent’s lifetime can also be extended. This occurs when patent term adjustments (PTA) and patent term extensions (PTE) are granted. IFI CLAIMS calculates and publishes an “Adjusted Expiration Date” based on these. IFI considers USPTO PTAs, PTEs and FDA Orange Book data.
For example: US-6740669-B1:

Application Date: June 8, 1998
Standard Expiration Date: June 8, 2018 (based on the 20 year rule)
Patent Term Adjustment: 801 Days (based on 35 USC 154 – appears on first page)
Patent Term Extension: 819 Days (based on 35 USC 156 and captured by IFI, but not appearing on published document)
Total Extension: 1620 Days
IFI Adjusted Expiration Date: November 14, 2022.
These extensions are very important in the pharmaceutical industry, but can also apply in other technical areas.
There are other ways the expiration date can change. A Terminal Disclaimer can shorten the patent's term and a Supplementary Protection Certificate can be granted to extend it. Depending on the country, these changes may be complicated. Usually, it is not esy to get current and detailed information. Therefore, IFI does not consider them when computing the Adjusted Expiration Date. However, we do provide a flag indicating when they are present for a patent. When applicable, the “regulatory-extension” flag will indicate “yes”. For example:
<ifi-patent-status anticipated-expiration="20280207" regulatory-extension="yes">
The presence of a terminal disclaimer is also noted in the data. For example, US-9723102-B2:

Application filed: March 24, 2016, but first application was July 1, 2009.
Standard Expiration Date: July 1, 2029 (20 years from first application filing).
Term Extension: 24 days.
IFI Adjusted Expiration Date: July 25, 2029. An additional tag: terminal-disclaimer="yes" is included in the data.
As noted above, the Legal Status Indicator and Adjusted Expiration Date are useful estimates, but should not be considered the final legal ruling. This is especially true when terminal disclaimers and regulatory extensions exist. Litigation and actions such as appeal board reviews may also affect the legal status of a patent. IFI has a blog post on this topic at The USPTO's is very busy - and why it matters.
IFI Expiration Date Adjustments only impact a small number of patents. For US Grants with application dates in the year 1998, only 1.3% have expiration date adjustments. The majority of the grants expire before or during the year 2018. For the 1.3%, the graph below shows the number of US grants that expire in each year after 2018 (US Grants with applications filed in 1998). Expirations before 2018 are not shown even when there are extensions.
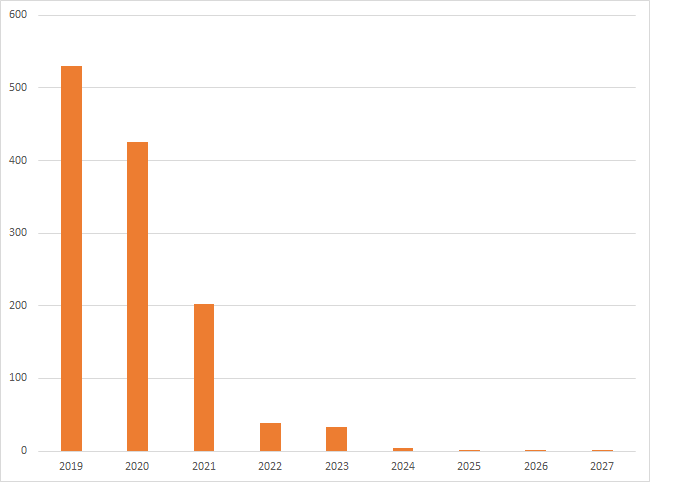
Patent expirations for patent applications filed in 1998.
Patent Application Status
IFI also publishes the status of patent applications. The possible values for published applications are:
- Pending
- Withdrawn
- Abandoned
- Granted
The current application status for the 317,855 published US applications with application dates in 2008 is shown below:
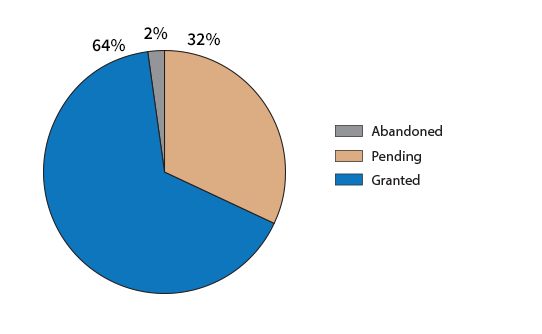
State of US applications filed in 2008.
It is interesting that 32% of the applications filed in 2008 are still pending. An example is:
US-20160128679-A1:
Published May 12, 2016: US application filed January 25, 2008, "HAEMOSTAT APPLICATION" by Medtrade Products Ltd. (UK).
IFI CLAIMS has a blog post on this topic: Korean Patent Office Leads in 2015 Patent Pendency, EPO and DE Lag .
Some patent applications are abandoned after pre-publication, but before grant. The number of these is small. But, you cannot assume that because a patent application has been published that a grant will inevitably follow.
The technical information disclosed in a patent is relevant to prior art searchers even when the patent is not in force. But for Freedom to Operate decisions, Mergers and Acquisition or Licensing negotiations, the legal status of a patent is critical. CLAIMS Direct subscribers have access to data that can be searched and reported on to make this work easier.
If you have any questions on our specific rules, data or country coverage, please contact us at info@ificlaims.com.